

The CPI is positioned as an independent institution focused on advancing pharmaceutical innovation research, conducting comprehensive analyses of pharmaceutical R&D and innovation. Its findings, highly relevant to both academia and pharmaceutical practice, are published in leading drug discovery journals.
Our interdisciplinary team of academics and industry professionals is committed to advancing scientific research in R&D management and innovation. Our aim is to support leading companies in overcoming the R&D productivity gap, thereby helping to maintain the affordability of healthcare systems.

Our work and publications in recent years have garnered significant attention from both industry and academia. Pharma executives, consultants, private equity investors, and academic researchers have reached out to us to discuss our research and findings and to address their questions. We believe that our topics and analyses have effectively captured the pulse of the times. Ultimately, our goal is to better understand pharma R&D and to share our knowledge with others to support their decision-making.
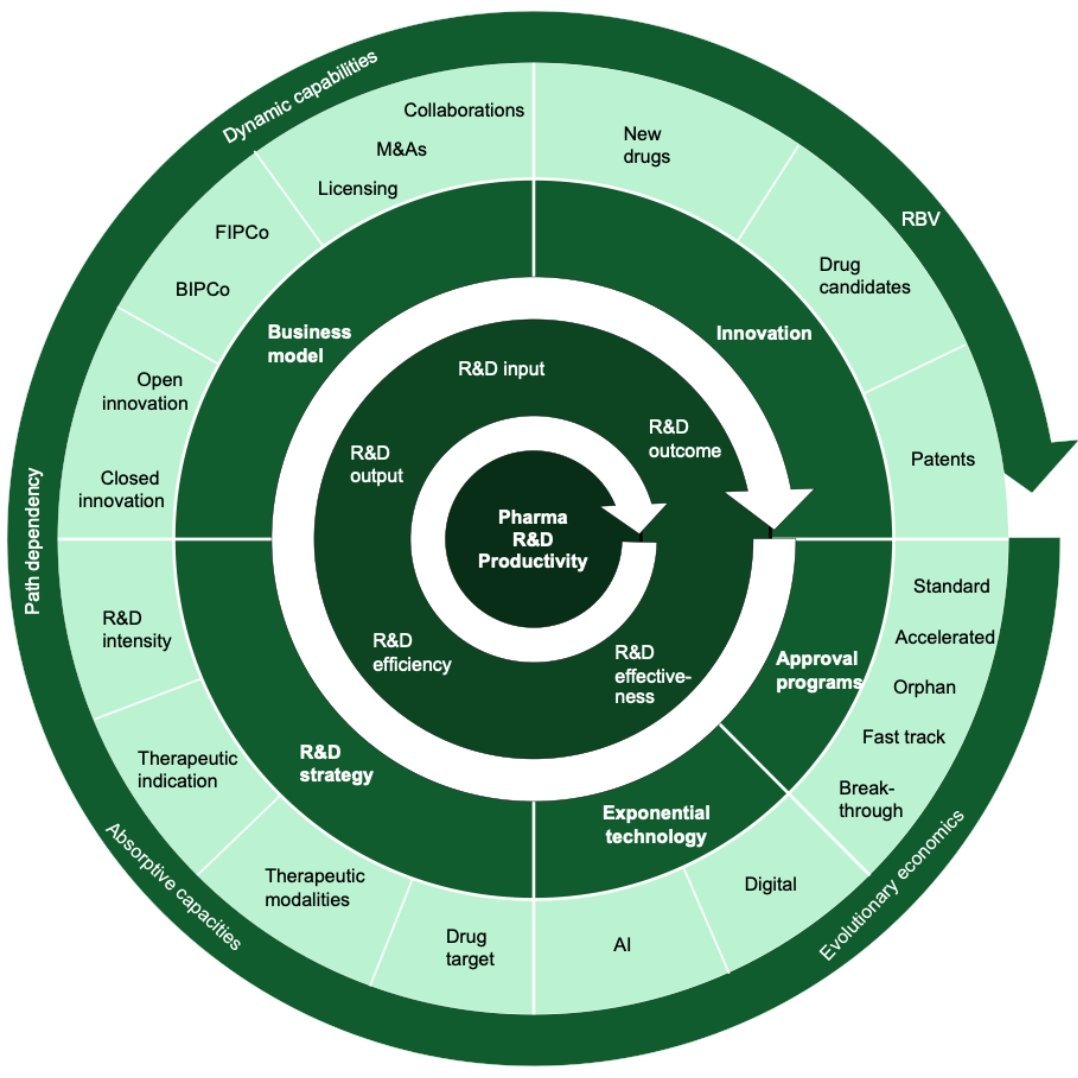
Pharmaceutical R&D is a complex endeavour. It is not just drug discovery and development or a simple gated process. As our perspective on pharma R&D is grounded in management and contextualized by the R&D productivity challenge, we understand that pharmaceutical R&D is fundamentally about leading innovation (Figure 1).
Our key fields of observation are
To put it in one sentence: It’s about better understanding how leading companies create and capture medical and commercial value.
We are consistently dedicated to publishing our latest insights in the leading scientific journals that are highly relevant to the pharmaceutical industry, such as Drug Discovery Today, Nature Reviews Drug Discovery, Journal of Translational Medicine, or Expert Opinion in Drug Discovery (Figure 2). Our work has also been cited or featured in the press, including German-language newspapers and publications such as Forbes.
2025
Schuhmacher, A., Hinder, M., Brief, E., Gassmann, O. & Hartl, D. (2025). Benchmarking R&D success rates of leading pharmaceutical companies: an empirical analysis of FDA approvals (2006–2022). Drug Discovery Today, 104291. doi:10.1016/j.drudis.2025.104291.
2024
Schuhmacher, A. (2024). Pharma innovation: how evolutionary economics is shaping the future of pharma R&D. Drug Discovery Today, 29(12), 104222. doi: 10.1016/j.drudis.2024.104222.
Schuhmacher, A. (2024). Exploring open source as a strategy to enhance R&D productivity. Expert Opinion On Drug Discovery, 1–4. doi: 10.1080/17460441.2024.2417352.
Gassmann, O., Schuhmacher, A., Von Zedtwitz, M. & Reepmeyer, G. (2024). Führung und Organisation pharmazeutischer Innovation. doi: 10.1007/978-3-031-43818-9.
Schuhmacher, A., Gassmann, O., Hinder, M. & Hartl, D. (2024). Comparative analysis of FDA approvals by top 20 pharma companies (2014–2023). Drug Discovery Today, 29(9), 104128. doi: 10.1016/j.drudis.2024.104128.
2023
Schuhmacher, A., Hinder, M., Boger, N., Gassmann, O. & Hartl, D. (2023). Is the blockbuster imperative broken? Drug Discovery Today, 28(11), 103789. doi: 10.1016/j.drudis.2023.103789.
Schuhmacher, A., Hinder, M., Von Stegmann und Stein, A., Hartl, D. & Gassmann, O. (2023). Analysis of pharma R&D productivity – a new perspective needed. Drug Discovery Today, 28(10), 103726. doi: 10.1016/j.drudis.2023.103726.
Schuhmacher, A., Hinder, M., Dodel, A., Gassmann, O. & Hartl, D. (2023). Investigating the origins of recent pharmaceutical innovation. Nature Reviews Drug Discovery, 22(10), 781–782. doi: 10.1038/d41573-023-00102-z.
2022
Schuhmacher, A., Hinder, M., Boger, N., Hartl, D. & Gassmann, O. (2022). The significance of blockbusters in the pharmaceutical industry. Nature Reviews Drug Discovery, 22(3), 177–178. doi: 10.1038/d41573-022-00213-z.
Schuhmacher, A., Haefner, N., Honsberg, K., Goldhahn, J. & Gassmann, O. (2022). The dominant logic of Big Tech in healthcare and pharma. Drug Discovery Today, 28(2), 103457. doi: 10.1016/j.drudis.2022.103457.
Schuhmacher, A., Gassmann, O., Bieniok, D., Hinder, M. & Hartl, D. (2022). Open innovation: A paradigm shift in pharma R&D? Drug Discovery Today, 27(9), 2395–2405. doi: 10.1016/j.drudis.2022.05.018.
2021
Schuhmacher, A., Brieke, C., Gassmann, O., Hinder, M. & Hartl, D. (2021). Systematic risk identification and assessment using a new risk map in pharmaceutical R&D. Drug Discovery Today, 26(12), 2786–2793. doi: 10.1016/j.drudis.2021.06.015.
Schuhmacher, A., Gatto, A., Kuss, M., Gassmann, O. & Hinder, M. (2021). Big Techs and startups in pharmaceutical R&D – A 2020 perspective on artificial intelligence. Drug Discovery Today, 26(10), 2226–2231. doi: 10.1016/j.drudis.2021.04.028.
Schuhmacher, A., Wilisch, L., Kuss, M., Kandelbauer, A., Hinder, M. & Gassmann, O. (2021). R&D efficiency of leading pharmaceutical companies – A 20-year analysis. Drug Discovery Today, 26(8), 1784–1789. doi: 10.1016/j.drudis.2021.05.005.
Hartl, D., De Luca, V., Kostikova, A., Laramie, J., Kennedy, S., Ferrero, E., Siegel, R., Fink, M., Ahmed, S., Millholland, J., Schuhmacher, A., Hinder, M., Piali, L. & Roth, A. (2021). Translational precision medicine: an industry perspective. Journal Of Translational Medicine, 19(1). doi: 10.1186/s12967-021-02910-6.
Schuhmacher, A., Gassmann, O., Hinder, M. & Kuss, M. (2020). The present and future of project management in pharmaceutical R&D. Drug Discovery Today, 26(1), 1–4. doi: 10.1016/j.drudis.2020.07.020.
Before 2021
Schuhmacher, A., Gatto, A., Hinder, M., Kuss, M. & Gassmann, O. (2020). The upside of being a digital pharma player. Drug Discovery Today, 25(9), 1569–1574. doi: 10.1016/j.drudis.2020.06.002.
Schuhmacher, A. & Kuss, M. (2020). The impact of crowdsourcing in modern drug discovery. Expert Opinion On Drug Discovery, 15(8), 865–867. doi: 10.1080/17460441.2020.1751116.
Schuhmacher, A., Gassmann, O., Kuss, M. & Hinder, M. (2019). The Art of Virtualizing Pharma R&D. Drug Discovery Today, 24(11), 2105–2107. doi: 10.1016/j.drudis.2019.07.004.
Schuhmacher, A., Gassmann, O., McCracken, N. & Hinder, M. (2018). Open innovation and external sources of innovation. An opportunity to fuel the R&D pipeline and enhance decision making? Journal Of Translational Medicine, 16(1). doi: 10.1186/s12967-018-1499-2.
Gassmann, O., Schuhmacher, A., Von Zedtwitz, M. & Reepmeyer, G. (2018). Leading pharmaceutical innovation: How to Win the Life Science Race. Springer.
Schuhmacher, A., Gassmann, O. & Hinder, M. (2016). Changing R&D models in research-based pharmaceutical companies. Journal Of Translational Medicine, 14(1). doi: 10.1186/s12967-016-0838-4.
Schuhmacher, A., Germann, P., Trill, H. & Gassmann, O. (2013). Models for open innovation in the pharmaceutical industry. Drug Discovery Today, 18(23–24), 1133–1137. doi: 10.1016/j.drudis.2013.07.013.
Our aspiration is to collaborate with the best experts from academia and industry. For instance, the collaboration with Prof. Dr. Dominik Hartl (GraniteBio) and Prof. Dr. Markus Hinder (Novartis) particularly reflects our desire to work with pharmaceutical experts who, like us, are passionate about research and gaining new insights into pharma R&D (Figure 3). Our shared successes demonstrate that we are on the right path. Curiosity, creativity, a passion for research, and a high level of self-motivation drive us forward.